Molecular Weight: 131.13
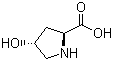
Molecular Formula: C5H9NO3
Canonical SMILES: C1C(CNC1C(=O)O)O
InChI: InChI=1S/C5H9NO3/c7-3-1-4(5(8)9)6-2-3/h3-4,6-7H,1-2H2,(H,8,9)/t3-,4+/m1/s1
InChIKey: PMMYEEVYMWASQN-DMTCNVIQSA-N
Boiling Point: 355.2±42.0°C at 760 mmHg
Melting Point: 274°C
Purity: 98%
Solubility: Soluble in Methanol (Slightly), Water (Sparingly)
Application:
L-Hydroxyproline, also known as 4-hydroxyproline, is a non-protein amino acid that plays a vital role in the structure and function of collagen, the most abundant protein in the human body. In recent years, researchers have been exploring the potential applications of L-Hydroxyproline in drug discovery and development due to its unique properties and biological activity.
First and foremost, it is critical to comprehend the significance of collagen in the human body as well as L-hydroxyproline's function in its manufacture. The primary structural element of many tissues, including as skin, bones, tendons, and cartilage, is collagen. It contributes to the general integrity and function of these tissues by giving them support, flexibility, and strength. One of the major amino acids found in collagen is hydroxyproline, which is necessary for the stability and correct folding of the triple helical structure of collagen. Collagen synthesis is hampered in the absence of L-hydroxyproline, which results in a number of connective tissue disorders.
Treating illnesses linked to collagen is one of the key therapeutic areas where L-hydroxyproline is being researched. These illnesses include Ehlers-Danlos syndrome, a collection of connective tissue disorders, osteogenesis imperfecta (brittle bone disease), and other hereditary illnesses affecting the synthesis or structure of collagen. Researchers hope to improve treatments for these disorders by creating new therapeutic techniques that target particular pathways of collagen production and modification, by better understanding the role of L-hydroxyproline in collagen formation.
L-hydroxyproline also has encouraging antioxidant and anti-inflammatory qualities, indicating that it may be used to create medications that address oxidative stress-related illnesses and inflammatory diseases. Numerous chronic illnesses, such as arthritis, cardiovascular disease, and neurological diseases, are characterized by inflammation. Compounds produced from L-hydroxyproline may provide novel treatment options for various disorders by targeting oxidative stress mechanisms and inflammatory pathways, which may improve patient outcomes and reduce symptoms.
Another area of interest for L-hydroxyproline applications is in the field of tissue engineering and regenerative medicine. Collagen-based biomaterials are commonly used in tissue engineering approaches due to their biocompatibility, biodegradability, and ability to promote cell adhesion and growth. By incorporating L-hydroxyproline into collagen-based scaffolds and matrices, researchers aim to enhance the mechanical properties and bioactivity of these materials, making them more suitable for applications such as wound healing, tissue repair, and organ regeneration.
In addition, L-hydroxyproline has shown potential as a targeted therapeutic drug delivery agent. Conjugating L-hydroxyproline to therapeutic agents or molecules can improve their stability, solubility, and bioavailability, thereby increasing their efficacy and reducing potential side effects. This approach may be particularly valuable in cancer treatment, where targeted drug delivery is essential to minimize off-target effects and increase therapeutic selectivity.
In addition to direct therapeutic applications, L-hydroxyproline is also a valuable tool for drug discovery and development. As a versatile building block, L-hydroxyproline can be used to design and synthesize novel small molecules, peptides, and peptidomimetics with specific biological activities. By incorporating L-hydroxyproline-containing motifs into drug candidates, researchers can modulate their pharmacokinetic and pharmacodynamic properties, thereby improving the potency, selectivity, and safety of the drug.
In addition, L-hydroxyproline can serve as a molecular probe to study protein-ligand interactions, protein folding, and enzymatic reactions. By labeling L-hydroxyproline with fluorescent or radioactive tags, researchers can track its uptake, distribution, and metabolism in biological systems, providing valuable insights into drug action and mechanisms of action. This approach is particularly useful in drug target validation, lead optimization, and drug metabolism studies where understanding how a drug candidate behaves in vivo is critical to successful development.
Storage: Store at 2-8°C under inert atmosphere
Synonyms: trans-4-Hydroxy-L-proline; L-4-hydroxyproline; trans-4-Hydroxyproline; H-Hyp-OH; hydroxy-L-proline; 4-Hydroxy-L-proline; trans-L-Hydroxyproline; 4-Hydroxy-2-pyrrolidinecarboxylic acid
Description
L-Hydroxyproline is an amino acid. It can be used as a nutrition and a flavor enhancer.
More details are to be found on supplier website